Register to receive a free Interventional Cardiology Devices Market Report Suite for U.S. synopsis and brochure
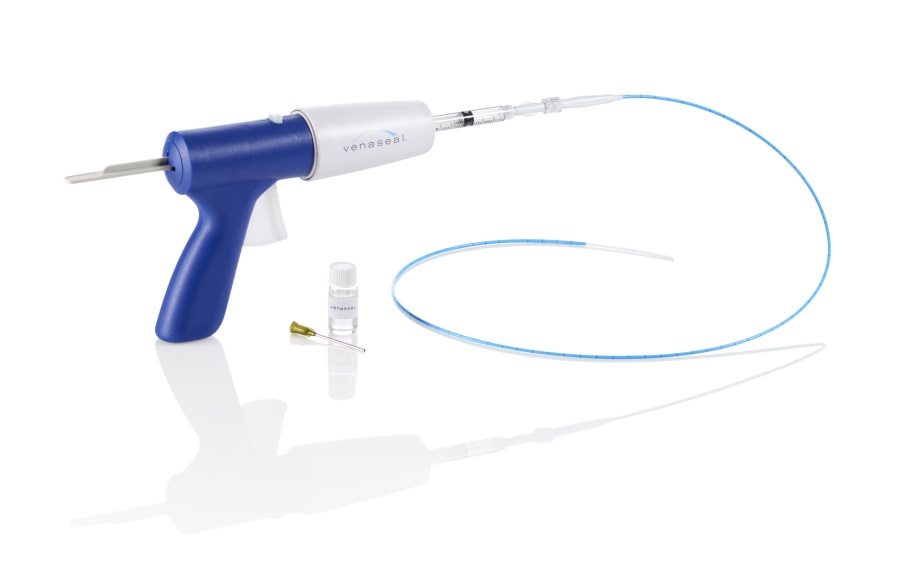
Medtronic has announced on July 25th that they have launched the VenaSeal closure system in India for patients with chronic venous disease. VenaSeal was engineered to be used in a minimally invasive procedure that uses a proprietary medical adhesive to close the diseased veins. Using imaging devices, the physician guides a catheter through a small incision in the leg and into the diseased area of the vein. Once the catheter is in the correct placement, the clinician administers the VenaSeal adhesive in segments and with manual compression, closes the vein. The blood is effectively re-routed through the other healthy veins in the leg.
“VenaSeal is an innovative approach to vein closure and is designed to minimize patient discomfort and reduce recovery time,” said Madan Krishnan, VP & managing director of India Medtronic in a news release. “Thousands of patients around the world have benefited from this therapy and we are pleased to now offer this advanced technology in India.”
This specialized methodology eliminates the risk of burning the vein or nerve injury which is more often associated with thermal-based venous procedures. The procedure is performed without the use of tumescent anesthesia, thus minimizing the need for multiple needle sticks. In the clinical trial, patients reported minimal to no ill effects, such as pain or bruising post procedure.
iData Research reports that similar to other medical device markets, the interventional cardiology market has been experiencing a period of consolidation in recent years. Large competitors are either entering the overall market or expanding their reach by entering new segments through strategic mergers and acquisitions. One trend that has risen as a result of consolidation is the bundling of devices at a discount. Bundling is driven by unfavorable reimbursement conditions and larger competitors, who have a wide variety of devices to offer across the interventional cardiology market and are able to negotiate more cost-effective contracts with hospitals and labs. As a result, competitive pressure is expected to contribute to declines in prices across the majority of segments in the market.
“Medtronic is committed to developing smart product and procedural solutions in the area of minimally invasive innovation to improve the quality of life for all patients,” said Madan Krishnan, vice president and managing director, India Medtronic Pvt Ltd. “VenaSeal is an innovative approach to vein closure and is designed to minimize patient discomfort and reduce recovery time”.
For Further Information
More on the interventional cardiology market in the U.S. can be found in a series of reports published by iData entitled the U.S. Market Report Suite for Interventional Cardiology