 Register to receive a free U.S. Market Report Suite for Infusion Therapy Device –MedSuite report synopsis and brochure
Register to receive a free U.S. Market Report Suite for Infusion Therapy Device –MedSuite report synopsis and brochure
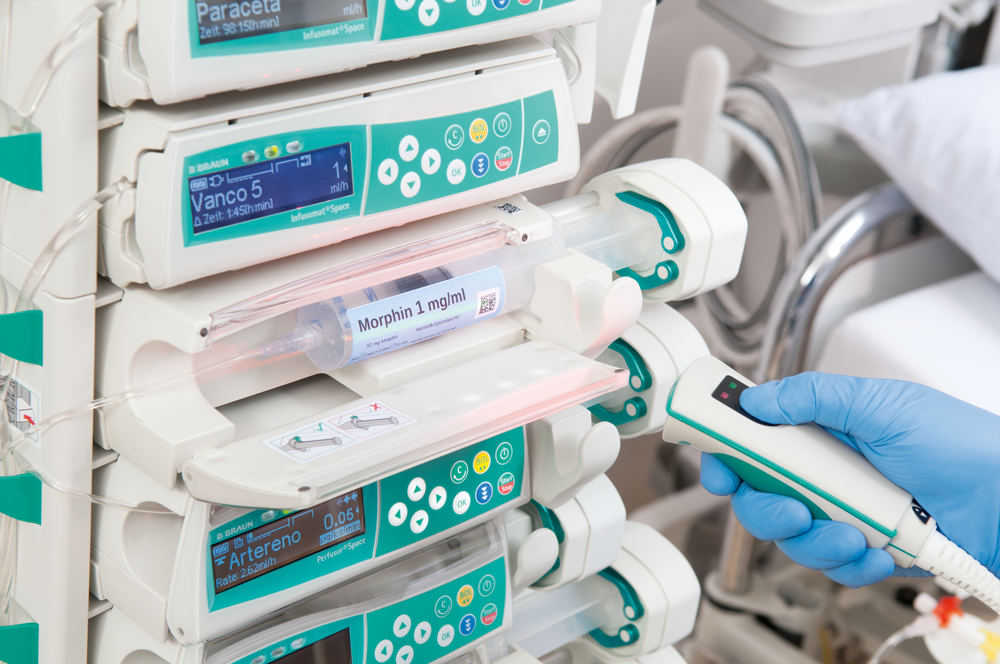
B. Braun Medical Inc., a leader in infusion therapy and pain management, recently premiered the wireless 2nd generation software for its Perfusor® Space Syringe Pump, at the 34th Annual National Association of Neonatal Nurses (NANN) conference in Anaheim, California.
The Perfusor® Space 2nd Generation Syringe Pump is the first syringe pump with air and road transport in the cleared indications for use. It is intended for use on adults, pediatrics and neonates for the intermittent or continuous delivery of parenteral fluids, enteral fluids, medications, blood and blood products through clinically accepted routes of administration. These routes include intravenous, intra-arterial, subcutaneous, epidural and enteral.
“This approval allows us to provide healthcare professionals a wireless syringe pump that offers complete interoperability and versatility across various care and transport settings,” said Tom Sutton, Vice President of Marketing, Medical Device and Pharmaceutical Systems at B. Braun Medical.
The Perfusor Space 2nd Generation Syringe Pump is the latest addition to the Synchronized Intelligence Infusion Platform. It offers the same user interface, wireless technology, and interoperability capabilities as the Infusomat® Space 2nd Generation Large Volume Pump. The Perfusor Space Syringe Pump also has complete integration with DoseTrac®, DoseGuard™, and AutoCOMPLETE™ Integrated EMR technology. This platform helps clinicians achieve 100% drug library compliance while minimizing medication errors and helping to reduce risk¹.
Key features of the pump include:
- – The first syringe pump with air and road transport in the cleared indications for use.
- – Wireless integration allows for complete interoperability.
- – Integrated piston brake helps prevent inadvertent bolus during syringe change.
- – Post-occlusion bolus reduction software designed to minimize bolus after occlusion.
- – Automatic drive technology designed for accuracy and safety.
- – A microprocessor in each pump allows for independent modularity, helping to prevent channel confusion and catastrophic pump failures.
- Compact design allows for vertical stacking in healthcare settings.
- – KeyGuard™ non-numeric keypad with simple arrows intended to prevent manual programming errors related to zero vs. decimal entry.
- – DoseGuard Drug Library Development Software, powered by FDB Infusion Knowledge™, creates customizable drug limits and clinical advisories with confidence.
- – DoseTrac Infusion Management Software allows for real-time pump infusion data reporting.
- – AutoCOMPLETE™ Integrated EMR that includes AutoProgramming, AutoDocumentation, – AutoNotification, and AutoLocation
For Further Information
More on the infusion therapy device market in the U.S. can be found in a series of reports published by iData entitled the U.S. Market Report Suite for Infusion Therapy Devices. The full report suite on the U.S. market for infusion therapy includes infusion pumps, intravenous (IV) sets, needleless connectors (NLCs) and stopcocks.
The iData series on the market for infusion therapy devices covers the U.S. and 15 countries in Europe including Germany, France, the United Kingdom (U.K.), Italy, Spain, Benelux (Belgium, Netherlands and Luxembourg), Scandinavia (Finland, Denmark, Sweden and Norway), Portugal, Austria and Switzerland. Reports provide a comprehensive analysis including units sold, procedure numbers, market value, forecasts, as well as detailed competitive market shares and analysis of major players’ success strategies in each market and segment. To find out more about infusion therapy device market data, register online or email us at info@idataresearch.net for a U.S. Market Report Suite for Infusion Therapy Devices brochure and synopsis.
 Register to receive a free
Register to receive a free