Product Description
The global pacemaker market was valued at $3.16 billion in 2025. This is expected to increase over the forecast period at a CAGR of 6.5% to reach over $4.9 billion.
The full report suite on the global market for pacemakers includes single-chamber pacemakers and dual-chamber pacemakers, each having transvenous and leadless markets.
The report includes data for 65 countries across 7 different regions which include North America, Latin America, Western Europe, Central & Eastern Europe, Middle East, Asia-Pacific, and Africa.
Data Types Included
- Unit Sales, Average Selling Prices, Market Size & Growth Trends
- Procedure Numbers
- Market Drivers & Limiters
- Market Forecasts Until 2032, and Historical Data to 2022
- Recent Mergers & Acquisitions
- Company Profiles and Product Portfolios
- Leading Competitors
Pacemaker Market Trends
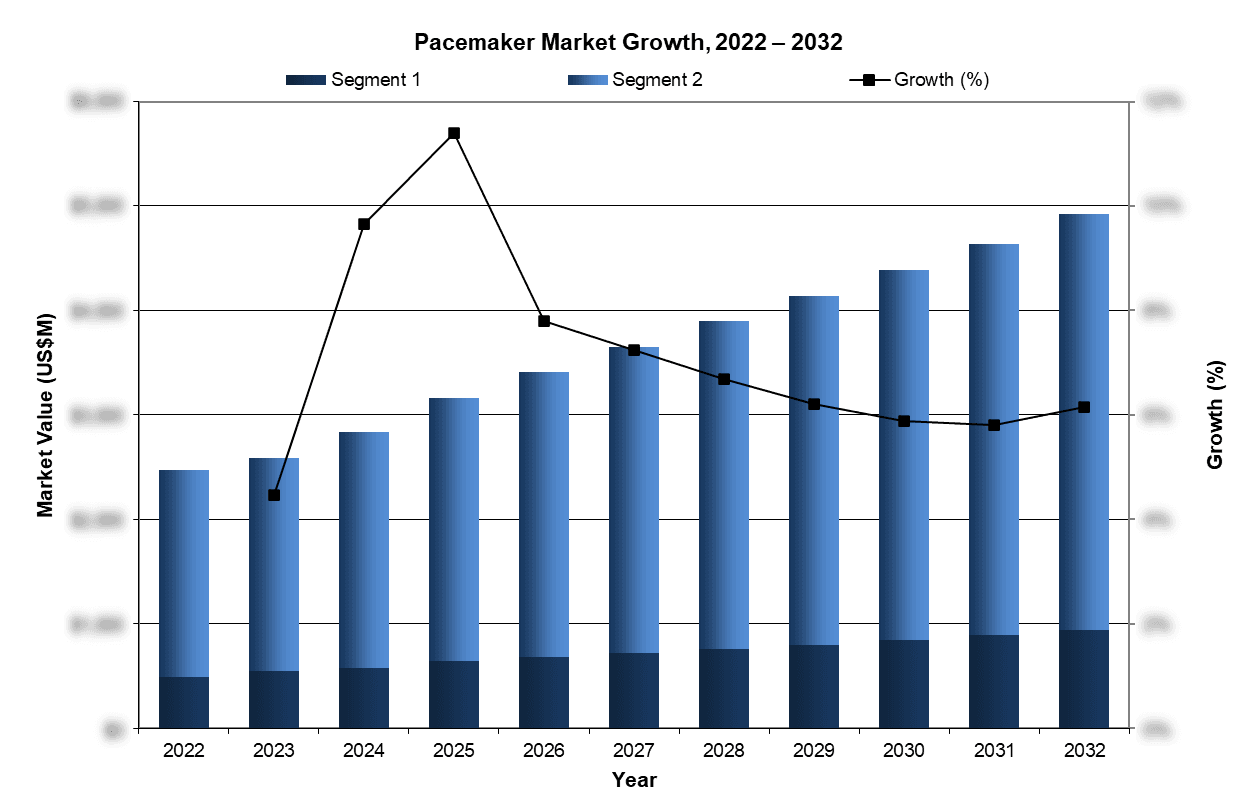
Current innovation is driven by advances in leadless pacing and increasingly sophisticated software that supports physiologic pacing and AI-assisted diagnostics. The rapid adoption of single-chamber leadless pacemakers has led to the steady decrease of the standard single-chamber pacemaker in key markets globally. Additionally, both standard and leadless dual-chamber pacemakers are experiencing healthy growth throughout the past years and are projected to continue to grow globally within the forecasted period. These developments are expected to slow the pace of price erosion by increasing the value of premium devices.
Pacemaker Market Share Insights

- Medtronic maintained the largest global share in the pacemaker market, effectively leading the market across all segments in 2025. The company’s competitive strength is anchored in its deep clinical evidence base and long-standing physician familiarity. Medtronic’s current lineup of pacemakers includes but is not limited to: Azure™, Advisa™ MRI SureScan™, Adapta™ and Attesta™. Medtronic also holds a dominant position in the leadless pacing segment since releasing the Micra™ family, and remains the most widely adopted platform globally, supported by extensive real-world registry data and high procedural uptake across major markets. Medtronic’s continued investment in physiologic pacing and device longevity further reinforces its leadership position as competition in both transvenous and leadless segments accelerates.
- Abbott is the second-leading competitor in the global pacemaker market, supported by its established Assurity MRI™ and Endurity™ platforms, as well as its expanding portfolio of next-generation leadless systems. The approval of the AVEIR VR leadless pacemaker in 2022 marked an important milestone, positioning Abbott as the second company to enter the leadless pacing space. Since then, the launch and rapid uptake of the AVEIR DR dual-chamber leadless system have become central to Abbott’s growth strategy. As the first modular dual-chamber leadless solution capable of direct atrial and ventricular communication, AVEIR DR is helping drive a shift toward fully leadless physiologic pacing. Strong early clinical adoption and expanding geographic approvals position AVEIR DR as a major long-term growth engine for Abbott, with the platform expected to play an increasingly significant role in market share expansion through 2026 and beyond.
- Boston Scientific is the third-leading competitor in the global pacemaker market, supported by its ACCOLADE™ and ESSENTIO™ MRI-compatible platforms and its VITALIO™, ADVANTIO™ and INGENIO™ legacy systems. While the company has maintained a solid presence in conventional transvenous pacing, its recent strategy has focused on steady portfolio refinement and integration with broader CRM offerings, including diagnostics, battery longevity improvements, and enhanced physiologic pacing capabilities. Although Boston Scientific does not currently participate in the leadless pacemaker segment, its established product families continue to serve as core contributors to the company’s CRM business heading into 2026.
Pacemaker Market Segmentation Summary
- Single-Chamber Pacemaker Market – Further Segmented Into:
- Lead Type: Transvenous and Leadless.
- Dual-Chamber Pacemaker Market – Further Segmented Into:
- Device Type: Transvenous and Leadless.
Research Scope Summary
| Report Attribute | Details |
|---|---|
| Regions | North America (Canada, United States) Latin America (Argentina, Bolivia, Brazil, Chile, Colombia, Costa Rica, Dominican Republic, Ecuador, Guatemala, Mexico, Panama, Paraguay, Peru, Puerto Rico, Uruguay, Venezuela) Western Europe (Austria, Benelux, France, Germany, Italy, Portugal, Scandinavia, Spain, Switzerland, United Kingdom) Central & Eastern Europe (Azerbaijan, Baltic States, Belarus, Bulgaria, Croatia, Czech Republic, Georgia, Greece, Hungary, Kazakhstan, Poland, Romania, Russia, Serbia, Slovakia, Slovenia, Turkey, Ukraine) Middle East (Bahrain, Iran, Israel, Kuwait, Oman, Qatar, Saudi Arabia, United Arab Emirates) Asia Pacific (Australia, Cambodia, China, Hong Kong, India, Indonesia, Japan, Kyrgyzstan, Macao, Malaysia, Mongolia, Myanmar, New Zealand, Pakistan, Philippines, Singapore, South Korea, Sri Lanka, Taiwan, Thailand, Uzbekistan, Vietnam) Africa (Algeria, Egypt, Ethiopia, Ghana, Kenya, Libya, Morocco, Nigeria, South Africa, Sudan, Uganda) |
| Base Year | 2025 |
| Forecast | 2026-2032 |
| Historical Data | 2022-2024 |
| Quantitative Coverage | Procedure Numbers, Market Size, Market Shares, Market Forecasts, Market Growth Rates, Units Sold, and Average Selling Prices. |
| Qualitative Coverage | Market Growth Trends, Market Limiters, Competitive Analysis & SWOT for Top Competitors, Mergers & Acquisitions, Company Profiles, Product Portfolios, FDA Recalls, Disruptive Technologies, Disease Overviews. |
| Data Sources | Primary Interviews with Industry Leaders, Government Physician Data, Regulatory Data, Hospital Private Data, Import & Export Data, iData Research Internal Database. |
CONTACT US FOR ADDITIONAL INFORMATION
For full segmentation and any questions regarding research coverage, please contact us for a complimentary demo of the full report.



